Groton, CT, November 22, 2021 – Astrocyte Pharmaceuticals Inc., a drug discovery and development company advancing novel neuroprotective therapeutics for the treatment of brain injuries, today announced the publication of a manuscript in the peer reviewed journal Stroke entitled: Adenosine A1R/A3R (Adenosine A1 and A3 Receptor) Agonist AST-004 Reduces Brain Infarction in a Nonhuman Primate Model of Stroke. The manuscript describes the results of pre-clinical studies using AST-004, Astrocyte’s lead drug candidate, for the treatment of stroke.
“According to the Centers for Disease Control (CDC) every 40 seconds someone in the United States has a stroke and every four minutes, someone dies as a result of a stroke,” said William Korinek, PhD, Chief Executive Officer, Astrocyte Pharmaceuticals. “This key study in non-human primates was designed to reproduce what typically happens when human stroke patients are treated in the emergency room. The results published today in Stroke show highly significant improvements across a number of metrics, with benefits beyond the current standard of care – reperfusion by thrombectomy surgery. Based on these data and the broad safety profile, we expect to initiate a Phase 1 clinical trial in the first half of 2022.”
The manuscript published in Stroke utilized a sophisticated non-human primate model of stroke with the study conducted by researchers from Astrocyte Pharmaceuticals and Hamamatsu Pharma Research, Inc. of Japan. Administration of AST-004 resulted in rapid and statistically significant decreases in lesion growth rate and total lesion volume. AST-004 treatment was well-tolerated with no observed adverse side effects. Researchers in the Department of Cell S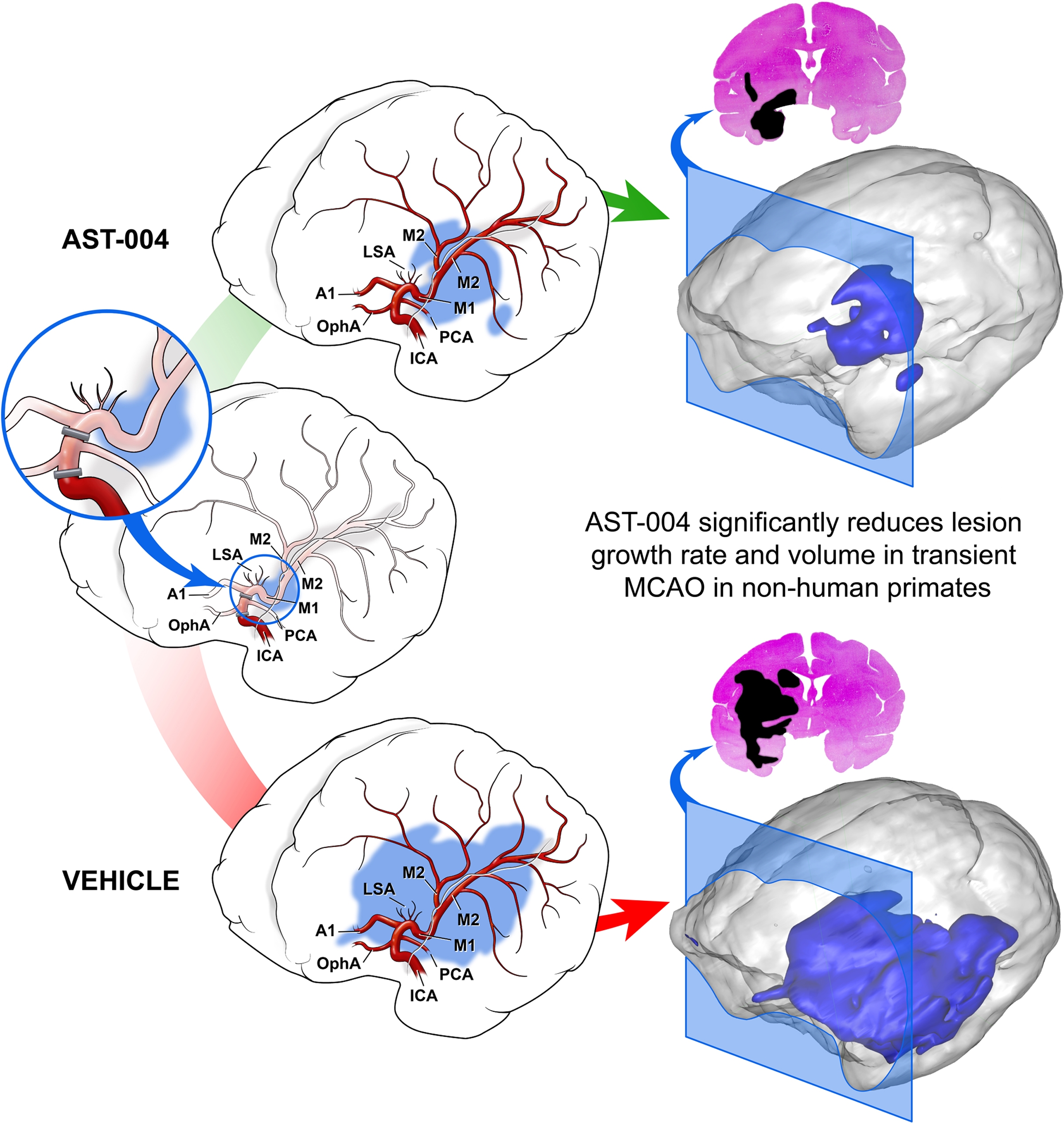 ystems and Anatomy at The University of Texas Health Science Center at San Antonio, and the Department of Neuroscience at the University of Warwick, United Kingdom collaborated on the research.
ystems and Anatomy at The University of Texas Health Science Center at San Antonio, and the Department of Neuroscience at the University of Warwick, United Kingdom collaborated on the research.
“Our novel approach focuses on maintaining energy metabolism in astrocytes, which are the natural caretaker cells of the brain and play a critical restorative role during brain stress or injury,” said Theodore E. Liston, PhD, Vice-President of Research, Astrocyte Pharmaceuticals and lead author. “The results published today in Stroke suggest that AST-004, our small molecule agonist that targets receptors on astrocytes, could significantly protect brain tissue during and after the ischemia experienced during a stroke. AST-004 administration changed the trajectory of the stroke, with lesion growth rates slowing by more than two-fold, and reducing the ultimate brain lesions by up to 45% beyond the benefits of reperfusion surgery alone. Treatment with AST-004 also preserved more of the penumbra brain tissue during the ischemia, which could potentially enable more stroke patients to reach hospitals and receive additional interventions like surgery.”
“AST-004 is a particularly exciting approach as it could potentially be used in a very wide range of stroke patients,” said Jeffrey L. Saver, MD, Director of the University of California Los Angeles Stroke Unit, and Chair of Astrocyte’s Clinical Advisory Board. “The mechanistic and safety profile of AST-004 could allow this treatment to be used pre-hospital, closer to the onset of a patient’s stroke, alongside a thrombolytic drug, and be utilized in stroke patients that both are or are not candidates for reperfusion surgery. If successful in future human clinical trials, AST-004 could dramatically change the way strokes are treated, improving the outlook for the approximately 13 million patients worldwide who suffer a stroke each year.”
###
About Astrocyte Pharmaceuticals Inc.
Astrocyte Pharmaceuticals Inc. is a privately held drug discovery and development company dedicated to accelerating the recovery and well-being of brain injury patients. The company is committed to proving the neuroprotective benefits of enhancing astrocyte function, and advancing breakthrough therapeutic agents for treating brain injury resulting from stroke, traumatic brain injury, concussion, and neurodegenerative disorders such as Alzheimer’s disease. For more information, please visit us at Astrocyte Pharmaceuticals Inc.